Important Medicine Safety Information
Important Medicine Safety Information
Jun 20, 2024
Recommended Calculations Of Contraception Duration After Completion Of Therapy To Minimise The Risk Of Embryotoxicity And Teratogenicity Associated With The Use Of Genotoxic Anticancer Medicines (Including Potential Metabolites)


The risk of acute tubulointerstitial nephritis (TIN) associated with proton pump inhibitors (PPIs)

GOLIMUMAB SIMPONI 50 MG: IMPORTANT CHANGES TO THE INSTRUCTIONS FOR USE (IFU) FOR THE SMARTJECT AUTOINJECTOR/PRE-FILLED PEN


IMBRUVICA (IBRUTINIB) – RISK OF FATAL CARDIAC EVENTS

COVID-19 VACCINES JANSSEN: RISK OF IMMUNE THROBOCYTOPENIA (ITP) AND VENOUS THROMBOEMBOLISM (VTE)




COVID-19 VACCINE JANSSEN: CONTRADICTION IN INDIVIDUALS WITH PREVIOUS CAPILLARY LEAK SYNDROME (CLS) & RISK OF THROMBOSIS IN COMBINATION WITH THROMBOCYTOPENIA



COVID-19 VACCINE JANSSEN: RISK OF THROMBOSIS IN COMBINATION WITH THROMBOCYTOPENIA


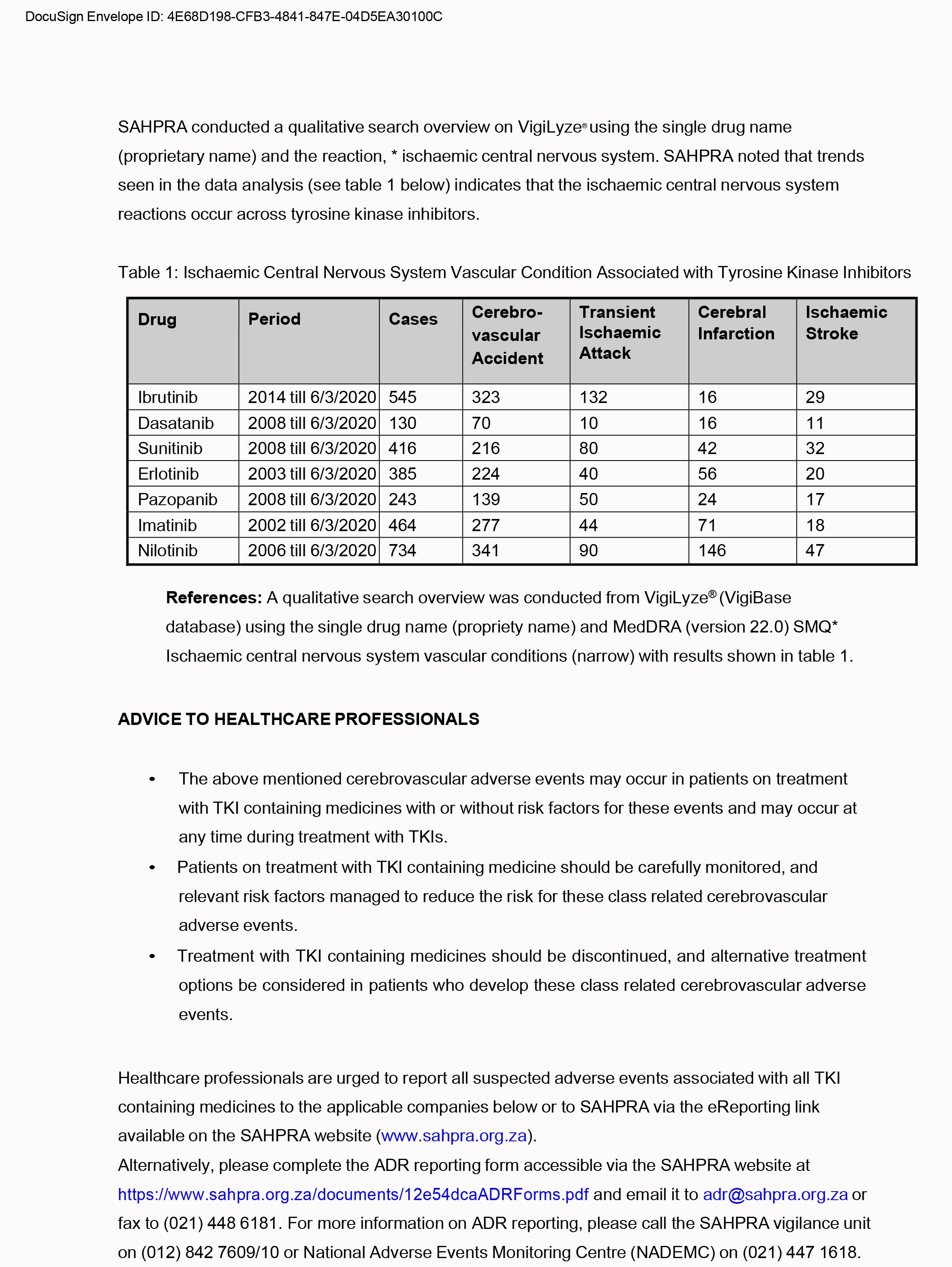
Warning about cerebro-vascular events, cerebro-vascular accident, cerebral infraction, ischaemic stroke and transcient ischaemic attack associated with the use of tyrosine kinase inhibitor containing medicines